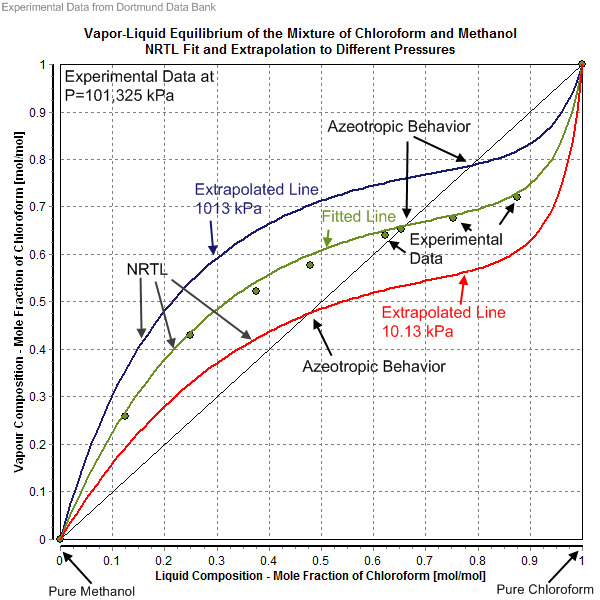
Processes | Free Full-Text | Experimental Data of Fluid Phase Equilibria- Correlation and Prediction Models: A Review

What Is the Optimal Activity Coefficient Model To Be Combined with the translated–consistent Peng–Robinson Equation of State through Advanced Mixing Rules? Cross-Comparison and Grading of the Wilson, UNIQUAC, and NRTL aE Models

Isobaric Binary and Ternary Vapor–Liquid Equilibrium for the Mixture of n-Hexane, Methylcyclopentane and N-Methylpyrrolidone | SpringerLink

What Is the Optimal Activity Coefficient Model To Be Combined with the translated–consistent Peng–Robinson Equation of State through Advanced Mixing Rules? Cross-Comparison and Grading of the Wilson, UNIQUAC, and NRTL aE Models

Thermodynamically consistent vapor-liquid equilibrium modelling with artificial neural networks - ScienceDirect

Thermo | Free Full-Text | Study of Thermodynamic Modeling of Isothermal and Isobaric Binary Mixtures in Vapor-Liquid Equilibrium (VLE) of Tetrahydrofuran with Benzene (303.15 K) Cyclohexane (333.15 K), Methanol (103 kPa), and Ethanol (100 kPa)

What Is the Optimal Activity Coefficient Model To Be Combined with the translated–consistent Peng–Robinson Equation of State through Advanced Mixing Rules? Cross-Comparison and Grading of the Wilson, UNIQUAC, and NRTL aE Models


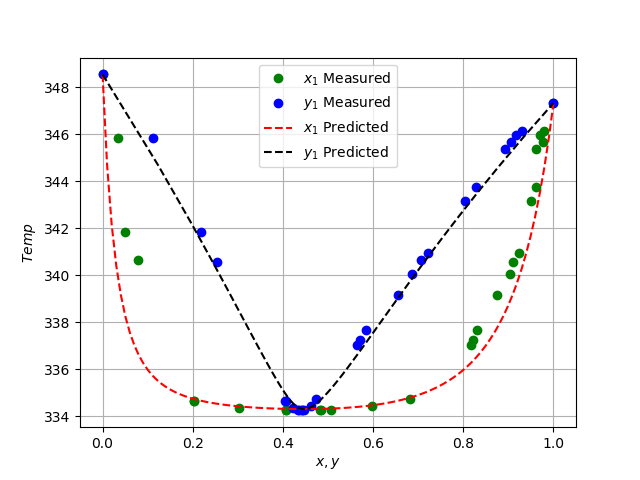

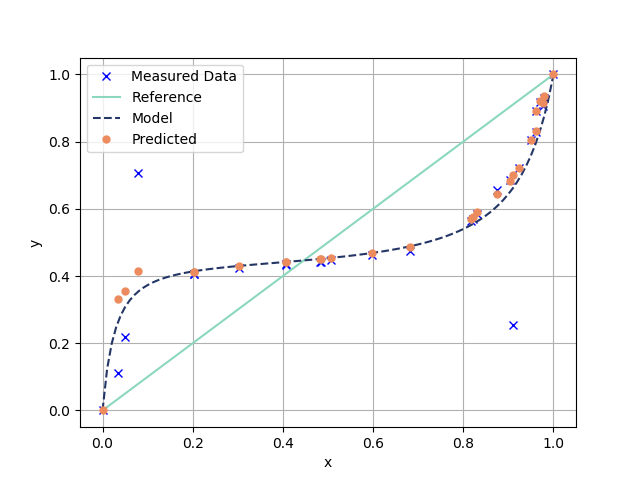
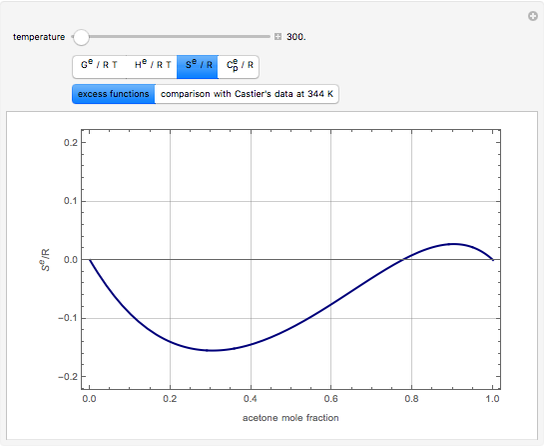



![PDF] Thermodynamics using Wilson and staggered quarks | Semantic Scholar PDF] Thermodynamics using Wilson and staggered quarks | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7e4fe768f9b286b482bf6a960a485226b4914a1a/5-Table1-1.png)